ZEISS expands ophthalmic surgical workflows in Mexico with COFEPRIS approval for ARTEVO 850 and MICOR 700
-
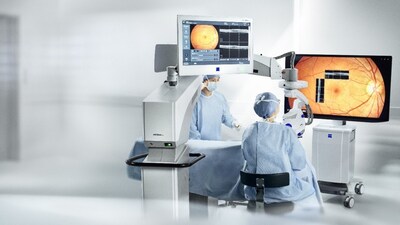
Elevates digital visualization for surgeons - ZEISS ARTEVO 850 surgical microscope provides ophthalmic surgeons in
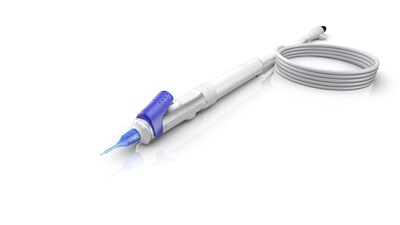
Mexico with advanced 3D visualization and end-to-end workflow integration. - Minimizes risk and increases O.R. efficiency for cataract surgery - ZEISS MICOR 700 first hand-held lens removal device with ultrasound-free operation reinvents lens extraction by providing a sustainable solution with a low initial investment to help cataract surgeons expand their intraocular working space.
JENA,
"ZEISS continues to expand its portfolio around the world, this time adding new surgical solutions in
"The ZEISS MICOR 700 and ZEISS ARTEVO 850 are true game changers in cataract surgery, elevating visualization and the workflow in the operating room. The introduction of these solutions into the Mexican market helps enable surgeons to operate with convenient control and efficiency," says
The ZEISS ARTEVO 850 3D heads-up ophthalmic microscope sets the pace in digital visualization with true color imaging and increased depth of field by nearly 60 percent.1 The surgical microscope also offers functional integration of devices and visualization technology to streamline clinical procedures. Digital integration into the ZEISS Ophthalmic Workflows allows for seamless data transfer. Additionally, the ZEISS ARTEVO 850 with CALLISTO eye® features a redesigned intuitive user interface that centralizes all controls on a single touchscreen.
The MICOR® 700 from ZEISS reinvents lens extraction with the first hand-held lens removal device with ultrasound-free operation, using the ZEISS NULEX (non-ultrasonic lens extraction) procedure to help surgeons broaden their intraocular working space, minimizing risk to surrounding eye structures and increasing operating room efficiency. The tip of the ZEISS MICOR 700 is designed to be gentle with the surrounding tissue. Its blunt tip design with rounded edges is designed to minimize the risk of tissue damage. ZEISS MICOR 700 also features a disposable "plug-and-play" system ensuring convenience, efficiency, and a minimal O.R. footprint.
For more information about ZEISS ophthalmic workflow solutions in
1 Data on file. Compared to ZEISS ARTEVO 800.
Not all products, services or offers are approved or offered in every market and approved labeling and instructions may vary from one country to another. For country-specific product information, see the appropriate country website. Product specifications are subject to change in design and scope of delivery as a result of ongoing technical development. The statements of the healthcare professionals reflect only their personal opinions and experiences and do not necessarily reflect the opinion of any institution that they are affiliated with. The healthcare professionals alone are responsible for the content of their experience reported and any potential resulting infringements.
Contact for investors
Director Investor Relations
Phone: +49 3641 220 116
Mail: investors.med@zeiss.com
Contact for the press
Head of
Ophthalmology,
Phone: +49 3641 220 331
Mail: press.med@zeiss.com
Brief Profile
The Group's head office is located in Jena,
For further information visit: www.zeiss.com/med
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/zeiss-expands-ophthalmic-surgical-workflows-in-mexico-with-cofepris-approval-for-artevo-850-and-micor-700-302770535.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/zeiss-expands-ophthalmic-surgical-workflows-in-mexico-with-cofepris-approval-for-artevo-850-and-micor-700-302770535.html
SOURCE