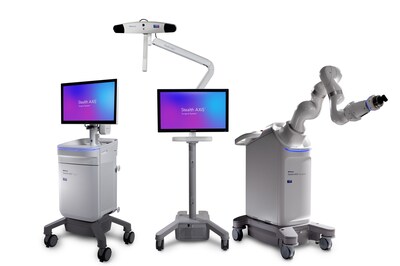
Medtronic receives FDA clearance for Stealth AXiS™ surgical system, first integrated planning, navigation and robotics platform for spine surgery
GALWAY, Ireland,
The Stealth AXiS™ system is cleared for spine procedures in
Grounded in more than 50 years of Medtronic leadership in surgical navigation and robotics, the Stealth AXiS™ system creates a clear pathway for adoption by combining familiar navigation workflows with a modular robotic design, allowing institutions to deploy what they need today and expand over time as clinical needs evolve.
"The Stealth AXiS™ system represents a meaningful step forward in how we think about navigation and robotics working together," said
A key innovation of the Stealth AXiS™ system is LiveAlign™ segmental tracking, an industry-first capability that allows surgeons to visualize anatomic motion, surgical adjustments, and patient alignment in real time during spine surgery, without the need for repeated imaging. This capability helps reduce reliance on manual steps and workflow disruption, supporting more consistent execution of patient-specific surgical plans.
As a cornerstone of Medtronic's AiBLE™ smart ecosystem, the Stealth AXiS™ system enables a more intuitive and seamless flow of information across the surgical continuum. By natively integrating planning, navigation, and execution within a single platform, the Stealth AXiS™ system helps streamline workflows and supports the exchange of insights before, during, and after spine surgery, allowing the broader AiBLE™ ecosystem to connect devices, software, and data in a more meaningful way.
"Spine surgery is complex, and variability remains a real challenge," said
The Stealth AXiS™ surgical system reflects Medtronic's focus on building durable platforms that simplify surgical workflows today while enabling future expansion across procedures, specialties, and care settings.
For more information about the Stealth AXiS™ surgical system and the AiBLE™ smart ecosystem, visit www.Medtronic.com/StealthAxis.
About the Cranial & Spinal Technologies Business at Medtronic
As the global market leader, Medtronic Cranial and Spinal Technologies is transforming the standard of care in spine and cranial surgery by putting patients first and addressing the complex challenges faced by spine and neurosurgeons. With a portfolio of 150 products covering more than 20 pathologies, we serve over 4 million patients annually. Building on a legacy of innovation, our AiBLE™ ecosystem integrates advanced technologies, data, and AI with a patient-centric approach, offering customizable solutions to enhance surgical precision, improve workflow efficiency, and achieve better outcomes, before, during, and beyond surgery.
About Medtronic
Bold thinking. Bolder actions. We are Medtronic.
Our technologies and therapies treat 70 health conditions and include cardiac devices, surgical robotics, insulin pumps, surgical tools, patient monitoring systems, and more. Powered by our diverse knowledge, insatiable curiosity, and desire to help all those who need it, we deliver innovative technologies that transform the lives of two people every second, every hour, every day. Expect more from us as we empower insight-driven care, experiences that put people first, and better outcomes for our world. In everything we do, we are engineering the extraordinary.
For more information on Medtronic, visit www.Medtronic.com and follow Medtronic on LinkedIn.
Any forward-looking statements are subject to risks and uncertainties such as those described in Medtronic's periodic reports on file with the Securities and Exchange Commission. Actual results may differ materially from anticipated results.
Contacts:
Public Relations
+1-269-377-2557
Investor Relations
+1-763-505-2696
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/medtronic-receives-fda-clearance-for-stealth-axis-surgical-system-first-integrated-planning-navigation-and-robotics-platform-for-spine-surgery-302687132.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/medtronic-receives-fda-clearance-for-stealth-axis-surgical-system-first-integrated-planning-navigation-and-robotics-platform-for-spine-surgery-302687132.html
SOURCE