GE HealthCare’s Photonova Spectra photon-counting CT receives FDA clearance
-
FDA clearance quickly follows Photonova Spectra’s debut at the
Radiological Society of North America (RSNA) 2025 Annual Meeting, marking rapid momentum for GE HealthCare’s innovative photon-counting CT technology - Photonova Spectra introduces the next generation of spectral photon-counting CT technology with 8‑bin energy resolution, powered by GE HealthCare’s proprietary Deep Silicon detector technology and a one‑scan universal workflow designed to boost efficiency and reduce complexity across a wide range of CT exams
-
Photonova Spectra is a result of the company’s
$5.1 billion innovation investment, leading to a wave of transformational products across the portfolio which combined are expected to drive 1-2% revenue growth
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260323095678/en/
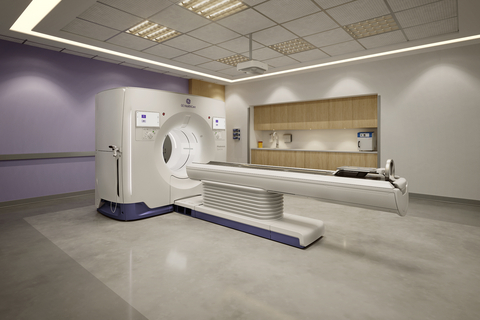
GE HealthCare’s Photonova Spectra photon-counting CT receives FDA clearance
Photon-counting CT represents a significant advancement in medical imaging. Unlike conventional CT systems – which first convert X-ray photons into visible light before measuring them – photon-counting CT directly counts individual photons and measures their energy. This approach enables higher spectral and spatial resolution as well as improved tissue characterization, helping provide clinicians with rich information to detect and diagnose disease with confidence.
“As clinicians across
Today, Photonova Spectra stands out with the introduction of Deep Silicon, a novel detector material designed to enhance spectral imaging performance. Leveraging the purity and structural consistency of silicon – a high-performing semiconductor material – Deep Silicon enables the precise measurement of photon energy and delivers high levels of energy resolution, which are critical for advanced image reconstruction. This capability can allow clinicians to obtain spectral images with high levels of contrast, combined with detailed visualization across neurological, oncological, musculoskeletal, thoracic, and cardiac imaging.
Furthermore, Deep Silicon with 8-bin energy resolution supports advanced material separation and characterization capabilities. This enables Photonova Spectra to clearly distinguish between different materials such as iodine, calcium and fat with remarkable precision. Its wide detector coverage and rapid rotation speed (0.23 seconds) also support fast acquisition and motion-free imaging – even in challenging patient scenarios.
Photonova Spectra automatically captures both 8-bin spectral and ultra-high definition spatial data simultaneously, without special setup or multiple protocols. This design gives clinicians access to spectral information in every exam, supporting confident decision-making and treatment monitoring in complex cases across specialties.
“Elevating diagnostic confidence, particularly with subtle low-contrast structures, requires increasing clarity significantly to facilitate enhanced material differentiation,” shares
To process the increased data volumes produced by photon‑counting CT, Photonova Spectra incorporates NVIDIA accelerated computing technology. Designed to handle up to 50 times more data than conventional CT,iv the GPU‑powered architecture is intended to help maintain smooth, efficient workflows by leveraging NVIDIA’s high-performance computing platform and CUDA-optimized reconstruction to turn massive spectral datasets into timely, clinically actionable images.
Workflow efficiency is further supported by a one‑scan, universal full‑fidelity approach intended to reduce exam‑specific protocols and enable automated reconstruction of ultra‑high definition spectral images on demand. The CT ONE operator environment and automated features — including Auto Positioning — are designed to help improve consistency across
Altogether, Photonova Spectra’s advanced architecture offers new opportunities for research in quantitative imaging, tissue characterization, and spectral biomarker discovery. To this end,
-
UW–Madison (
Madison, Wisconsin ): Represents the firstU.S. clinical evaluation site of GE HealthCare’s silicon based photon-counting CT, where researchers explore continuous improvement opportunities, such as elevating image quality, reducing noise and motion artifacts, enhancing soft tissue contrast, and exploring new clinical applications across oncology, cardiology, neurology and other CT intensive specialties. -
Stanford Medicine (
Palo Alto, California ): Concentrates on both human subject and technical research efforts – specifically assessing reconstruction methods, optimizing image presentation workflows, and identifying potential pathology specific advantages such as ultra-high-resolution structural visualization, improved tissue characterization, and potential new biomarkers.
“Photonova Spectra reflects years of intentional design and close collaboration with clinicians, researchers and collaborators across the globe,” adds
Leveraging GE HealthCare’s platform architecture, Photonova Spectra also is purposefully designed for easy install and to fit into existing GE HealthCare CT ready rooms with minimal changes, preserving the foundational mechanical, electrical, and workflow design used across the Revolution family.
With
News of Photonova Spectra’s 510(k) clearance quickly follows GE HealthCare’s unveiling of the technology at the
For more information on GE HealthCare’s Photonova Spectra photon-counting CT system, visit gehealthcare.com.
About
Follow us on LinkedIn, Facebook, Instagram, or visit our website for our latest news and perspectives.
i Photonova Spectra is 510(k) cleared with the
ii
iii Sittig, Dean F., and
iv When compared to Revolution Apex Elite.
View source version on businesswire.com: https://www.businesswire.com/news/home/20260323095678/en/
GE HealthCare Media Contact
M +1 608 381 8829
margaret.steinhafel@gehealthcare.com
Source:
