GE HealthCare Announces First Patient Dosed in Phase 2/3 LUMINA Trial for Manganese-Based MRI Contrast Agent Under FDA Fast Track Designation, Further Advancing Its Innovation Pipeline of Novel Imaging Agents
- Mangaciclanol, if approved, could transform contrast-enhanced MR imaging by offering an alternative to - or even replacing - gadolinium-based MRI agents
- A manganese-based MRI contrast agent could address concerns with gadolinium-based contrast agents around retention in the body, security of supply, and the environment, with mangaciclanol’s clinical development under FDA Fast Track designation
- The program demonstrates GE HealthCare’s commitment to advancing novel imaging agents to address unmet patient needs
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260423262096/en/
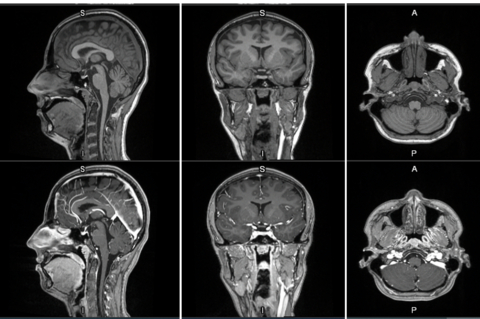
First-in-human contrast enhanced MRI images with
MRI contrast agents enhance the visualization of abnormal structures or lesions during imaging procedures and help clinicians better distinguish between healthy and diseased tissue. Mangaciclanol is intended for general-purpose MR imaging and demonstrates comparable relaxivity (the ability to enhance signal intensity) to market-leading gadolinium-based agent, gadobutrol, with early clinical images suggesting similar diagnostic capability. Unlike gadolinium, which is a rare-earth metal, manganese is present in our food, and is an endogenous element, naturally occurring and autoregulated in the body. The macrocyclic ‘cage-like’ structure of mangaciclanol lessens the possibility of retention.
Approximately one-third of global MRI procedures require a contrast agent for effective diagnosis,1 with around 65 million gadolinium contrast enhanced procedures globally each year.2 As a rare-earth element, gadolinium supply is largely dependent on mining and processing infrastructure in
Dr
“As demand for diagnostic imaging continues to rise, we continue to advance our imaging agent pipeline to better meet the needs of patients,” said
Phase 1 results for mangaciclanol showed the investigational agent was well tolerated in a first in human trial with no serious adverse events, no dose limiting toxicities, nor clinically relevant findings reported.
GE HealthCare’s
Mangaciclanol is in clinical development and currently not approved for use.
About
Follow us on LinkedIn, Facebook, Instagram, or visit our website for our latest news and perspectives.
1
Contrast Agents of Magnetic Resonance Imaging and Future Perspective.
Nanomaterials. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC10343825/[europepmc.org]
2Recent Developments and Future Perspectives in Magnetic Resonance Imaging and Computed Tomography Contrast Media.Frontiers in Radiology. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC12871431/
View source version on businesswire.com: https://www.businesswire.com/news/home/20260423262096/en/
GE HealthCare Media Contact:
M: +44 7920 591370
david.j.morris@gehealthcare.com
Source:
