IBM and University Researchers Create a Never-Before-Seen Molecule and Prove its Exotic Nature with Quantum Computing
- Published today in Science, the discovery marks the creation and observation of the first molecule with a half-Möbius electronic topology.
- It shows how quantum computers can directly contribute to understanding complex molecular behavior.
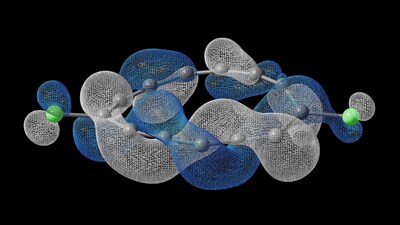
To the scientists' knowledge, a molecule with such topology has never before been synthesized, observed, or even formally predicted. Understanding this molecule's behavior at the electronic structure level required something equally fundamental: a high fidelity quantum computing simulation.
The discovery advances science on two fronts. For chemistry, it demonstrates that electronic topology — the property governing how electrons move through a molecule — can be deliberately engineered, not merely found in nature. For quantum computing, it is a concrete demonstration of a quantum simulation doing what it was designed to do: representing quantum mechanical behavior directly, at the molecular scale, to produce scientific insight that would otherwise have remained out of reach.
"First, we designed a molecule we thought could be created, then we built it, and then we validated it and its exotic properties with a quantum computer," said
A Never-Before-Seen Molecule
The molecule, with the formula C₁₃Cl₂, was assembled atom-by-atom at
Experiments with scanning tunneling and atomic force microscopy, both techniques pioneered at
This half-Möbius topology is qualitatively distinct from any previously known molecule and can be reversibly switched between clockwise-twisted, counterclockwise-twisted and untwisted states — demonstrating that electronic topology is not a property to be discovered, but one that can now be deliberately engineered under specific conditions.
A Disruptive Scientific Tool: Quantum-Centric Supercomputing
The scientists in this experiment created a molecule that had never existed. Now they had to figure out why it worked, a task which challenged conventional computers. The electrons within C₁₃Cl₂ interact in deeply entangled ways — each influencing all the others simultaneously. Modeling that behavior requires tracking every possible configuration of those interactions at once, requiring computational demands that grow exponentially and can quickly overwhelm classical machines.
Quantum computers are different by nature because they operate according to the same quantum mechanical laws that govern electrons in molecules, and they can represent these systems directly rather than approximate them. They "speak" the same fundamental language as the matter they are built to study and that distinction, once largely theoretical, can now contribute to concrete scientific results.
This capability offers tremendous potential for quantum computers to support real-world experimentation with quantum-centric supercomputing workflows. By integrating quantum processing units (QPUs), CPUs, and GPUs, quantum-centric supercomputing allows complex problems to be broken into parts that are orchestrated and solved according to each system's strengths — achieving what no single compute paradigm can deliver alone.
Utilizing an
This achievement builds on
RESEARCHER QUOTES
Dr. Igor Rončević, paper co-author, Lecturer in Computational and Theoretical Chemistry at
"Chemistry and solid-state physics advance by finding new ways to control matter. In the second half of the 20th century, substituent effects were very popular. For example, researchers explored how the potency of a drug or the elasticity of a material changes if, for example, a methyl is replaced with chlorine. The turn of the century brought us spintronics, introducing electron spin as a new degree of freedom to play with, and transforming data storage. Today, our work shows that topology can also serve as a switchable degree of freedom, opening a new powerful route for controlling material properties.
"The non-trivial topology of this molecule, and the exotic behavior of many other systems, arises from interactions between their electrons. Simulating electrons with classical computers is very hard – a decade ago we could exactly model 16 electrons, and today we can go up to 18. Quantum computers are naturally well-suited for this problem because their building blocks – qubits – are quantum objects, which mirror electrons. Using
Dr.
"It is remarkable that the Lewis structure of C₁₃Cl₂ already indicates it is chiral, as confirmed by the experiment and quantum chemical calculations. It is also amazing that the enantiomers can be interconverted by applying voltage pulses from the probe tip."
Dr.
"I'm really excited to be part of a project where quantum hardware does real science, not just demos. It's fascinating that a tiny molecule can have such a complex electronic structure that is challenging to simulate classically, and is so twisted and strange that it almost twists your mind."
For more about this research, please read the blog: Quantum simulates properties of the first-ever half-Möbius molecule, designed by
About
For more information, visit https://research.ibm.com.
Media Contact:
Edlehr@us.ibm.com
dave.mosher@ibm.com


Photo -
https://mma.prnewswire.com/media/2927254/Half_M_bius.jpg
Photo -
https://mma.prnewswire.com/media/2927253/blogArt_half_M_biusMolecule.jpg
Logo -
https://mma.prnewswire.com/media/95470/ibm_logo.jpg
![]() View original content:https://www.prnewswire.co.uk/news-releases/ibm-and-university-researchers-create-a-never-before-seen-molecule-and-prove-its-exotic-nature-with-quantum-computing-302705828.html
View original content:https://www.prnewswire.co.uk/news-releases/ibm-and-university-researchers-create-a-never-before-seen-molecule-and-prove-its-exotic-nature-with-quantum-computing-302705828.html

